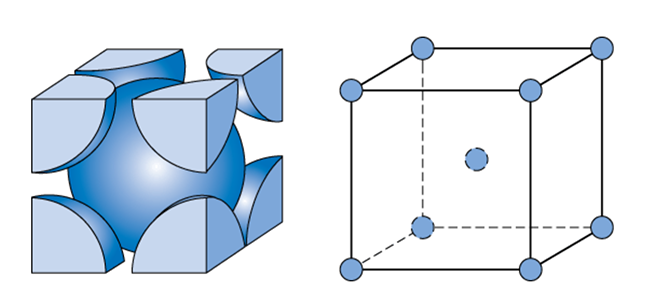
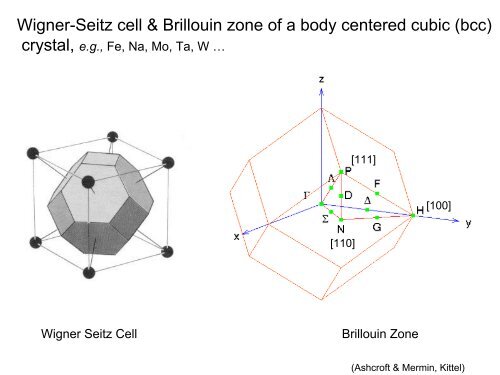
Crystal structures of (a) body-centered-cubic (bcc), (b) and (c) A15... | Download Scientific Diagram
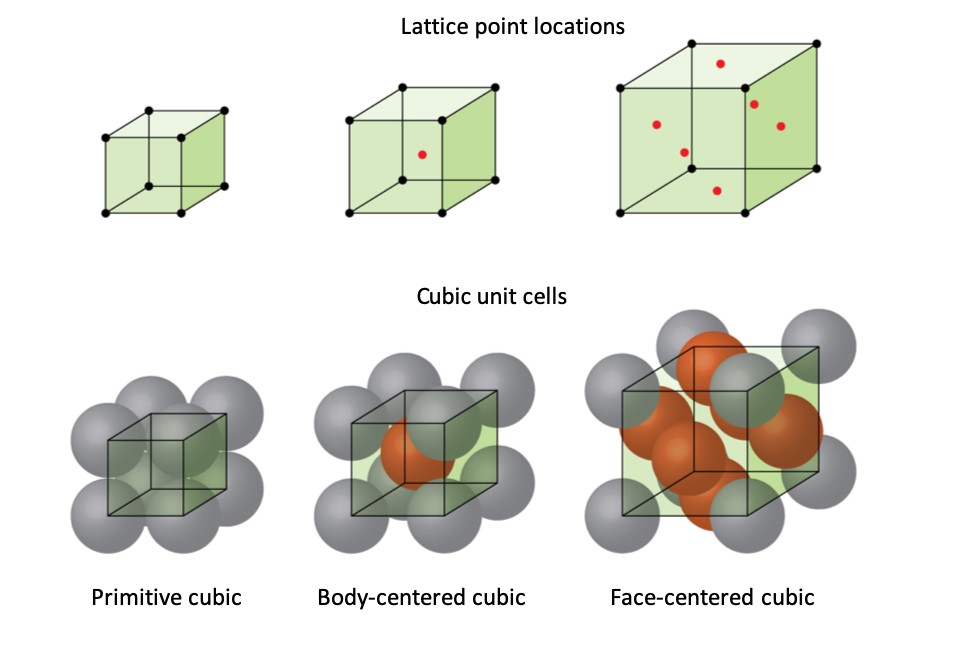
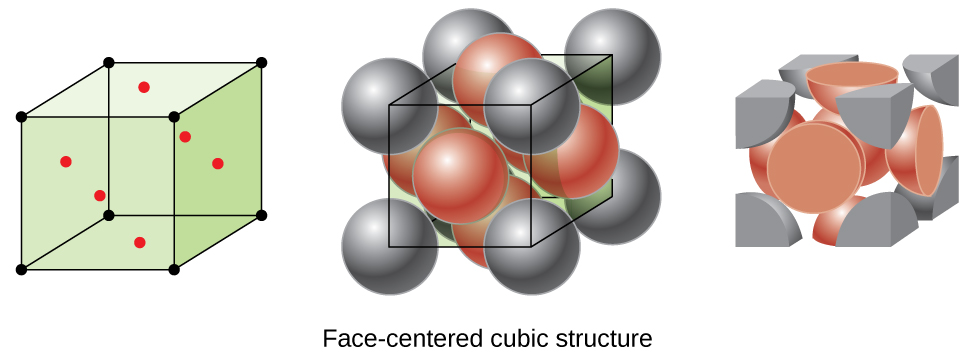
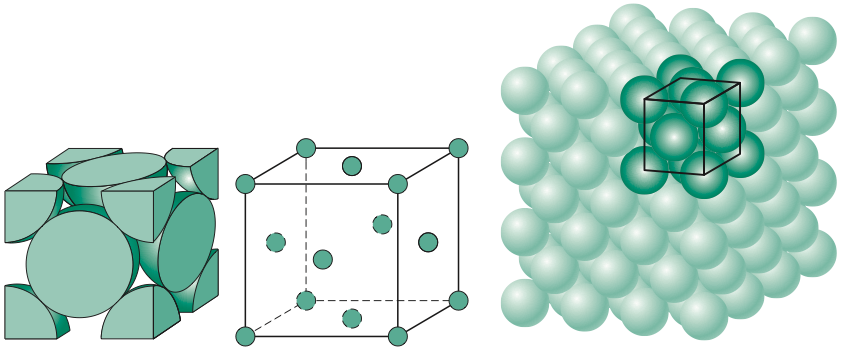
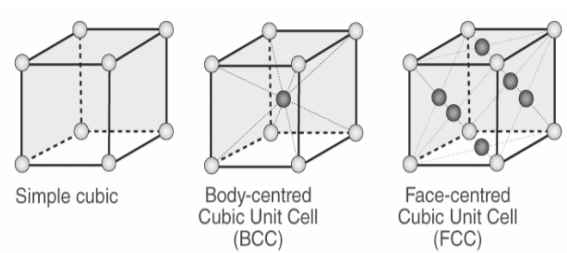
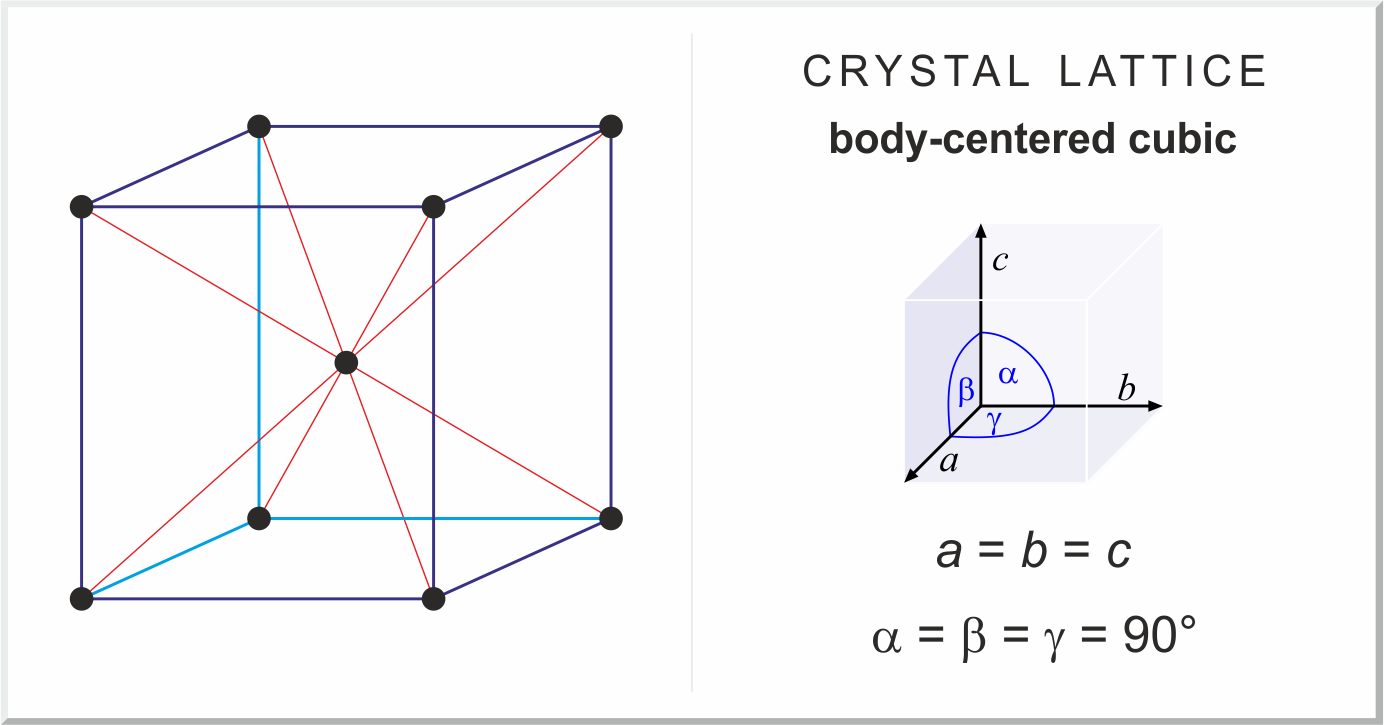
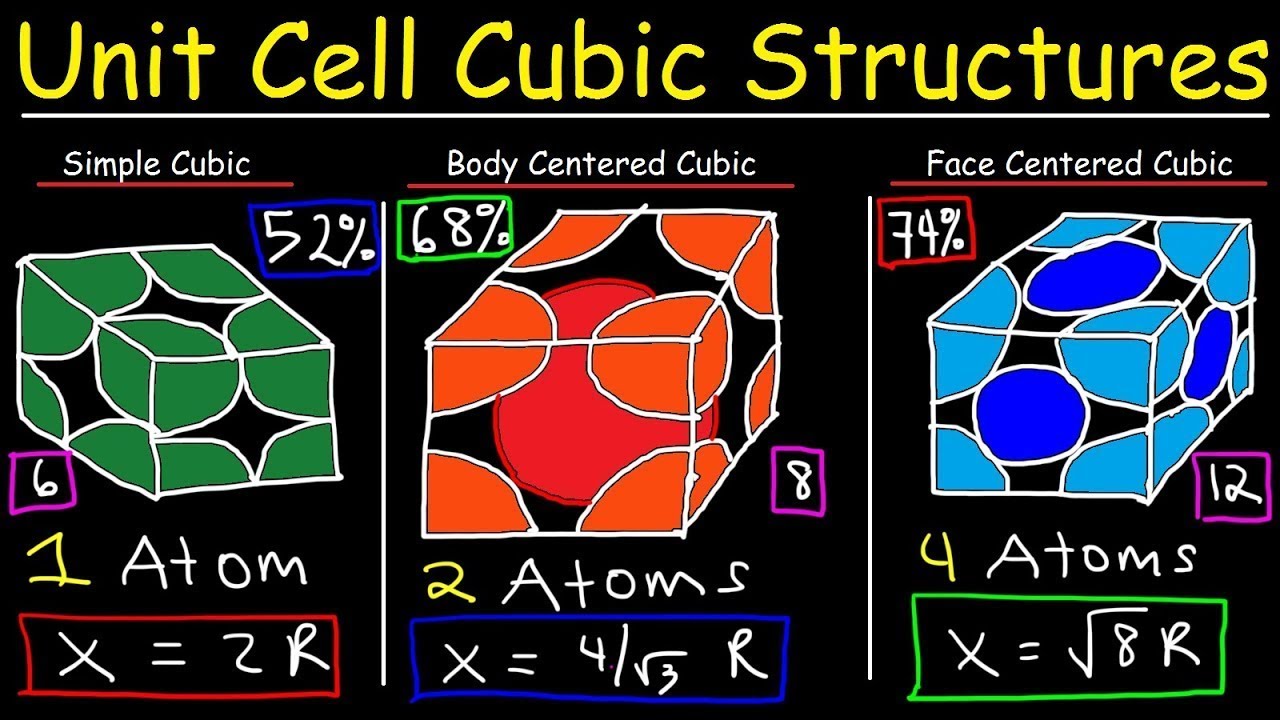
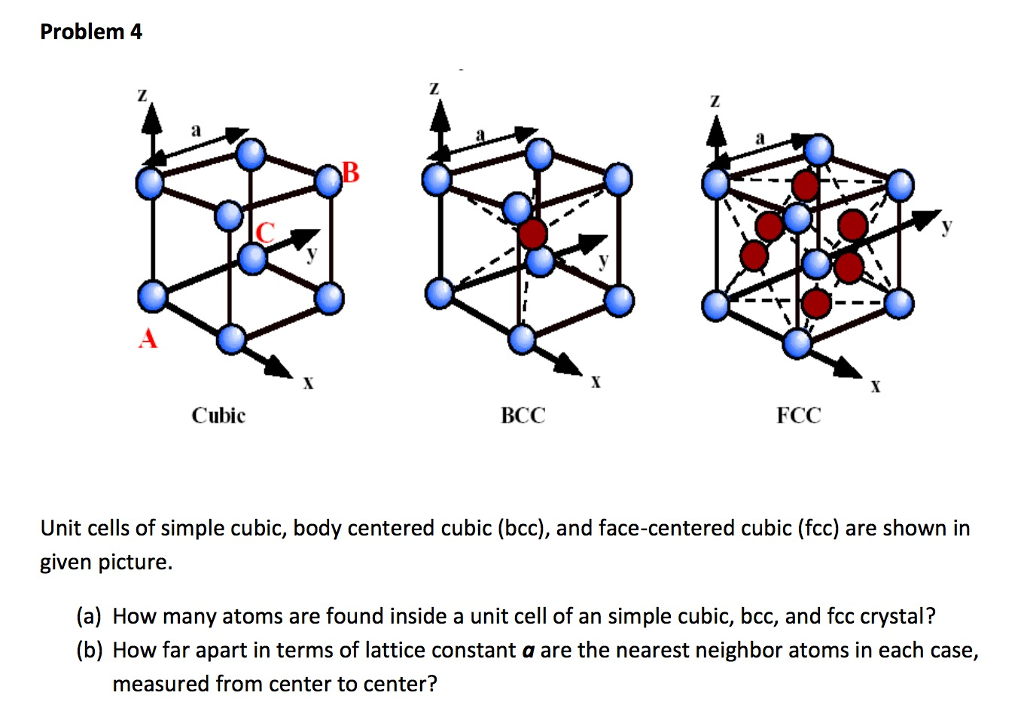
Types of Unit Cells: Body-Centered Cubic and Face-Centered Cubic (M11Q5) – UW-Madison Chemistry 103/104 Resource Book
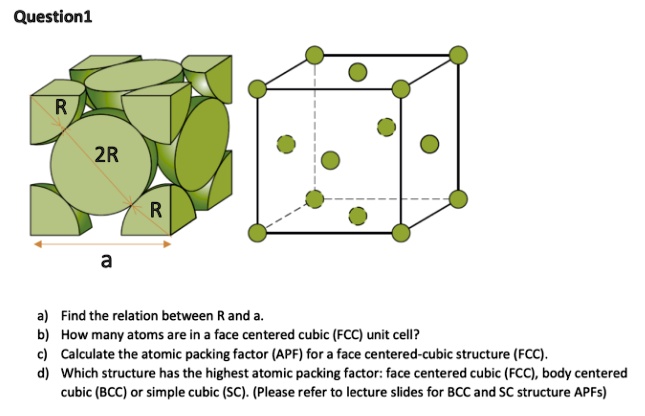
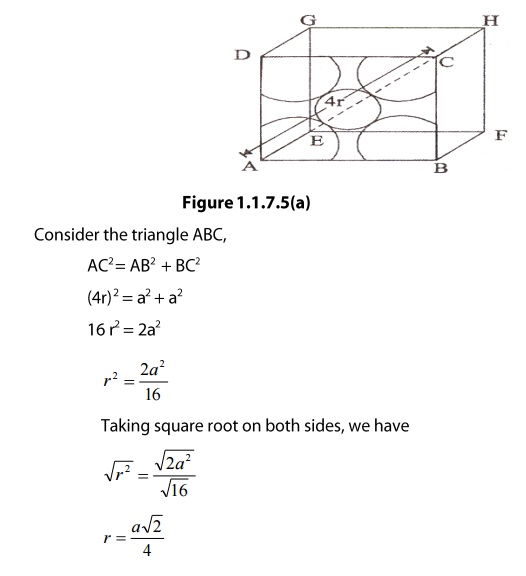
SOLVED: Question 1: Find the relation between R and how many atoms are in a face-centered cubic (FCC) unit cell. Calculate the atomic packing factor (APF) for the face-centered cubic structure (FCC).