
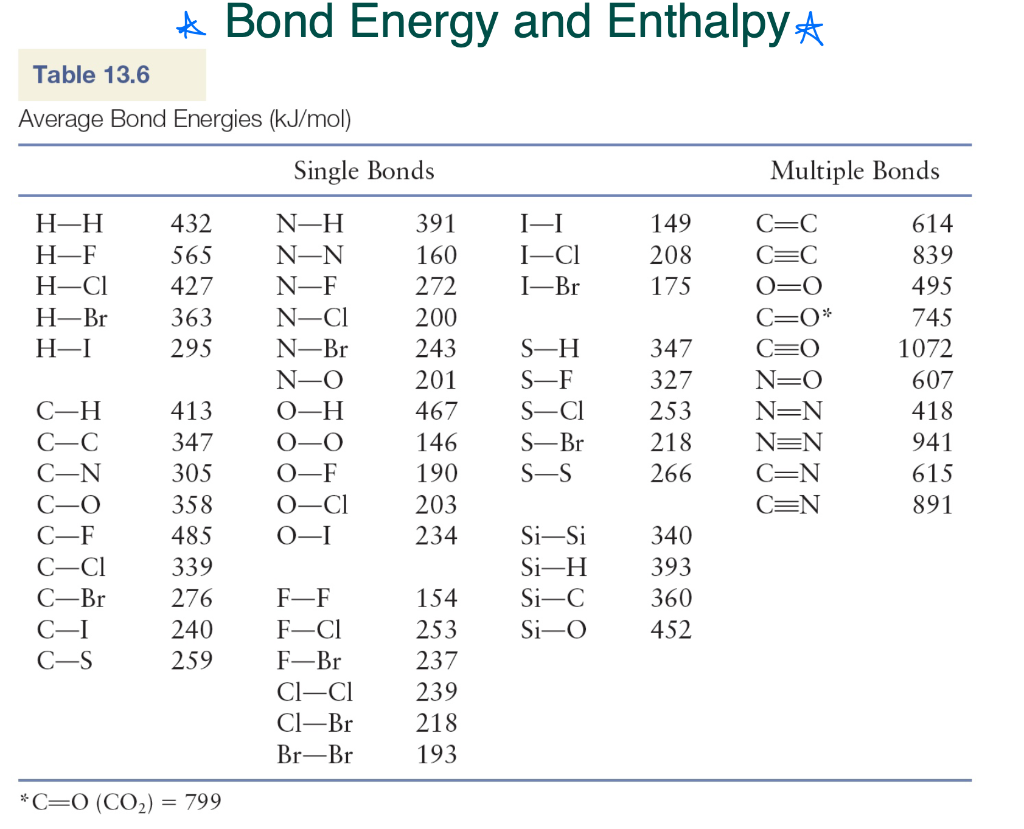
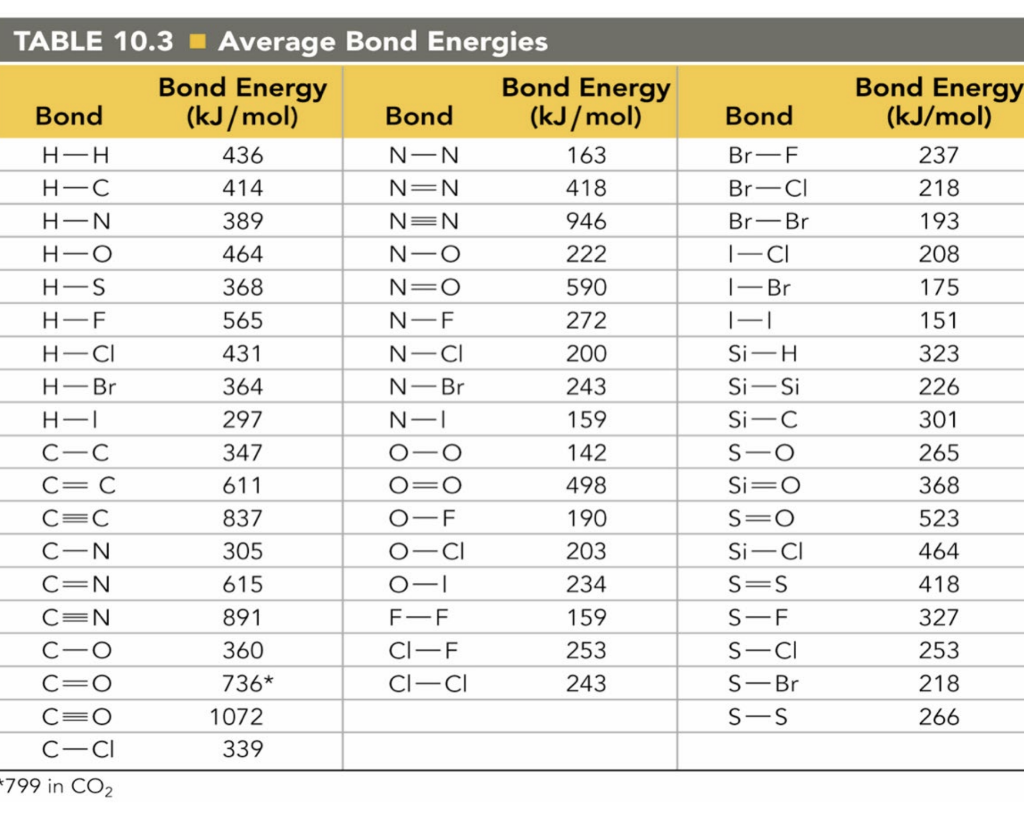
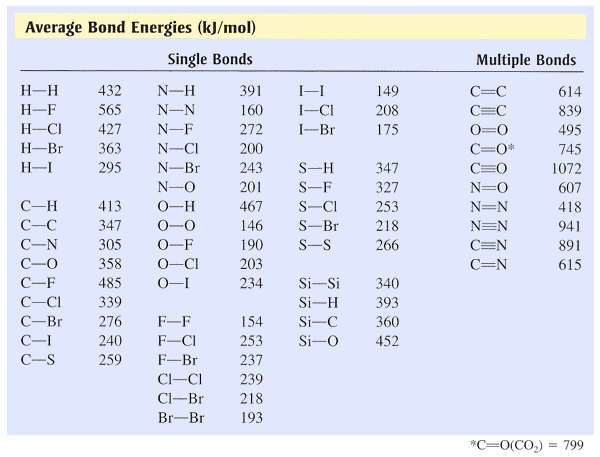
SOLVED: TABLE 13.6 Average Bond Energies (klmol) Single Bonds H-H 432 N-H 391 H-F 565 N-N 160 C-Cl 427 C-Br 363 H-C 295 N-Br 243 N-O 201 C-H 467 O-O 146 O-F
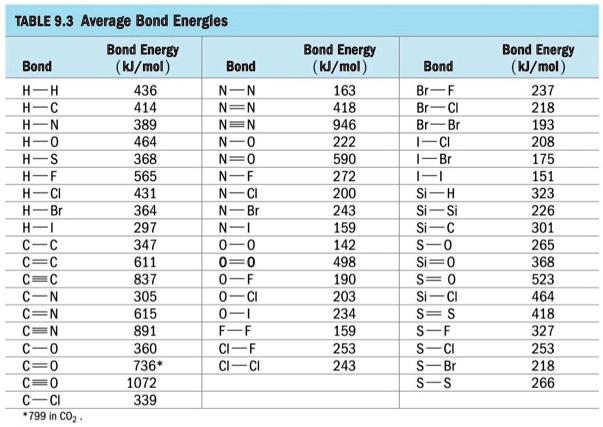
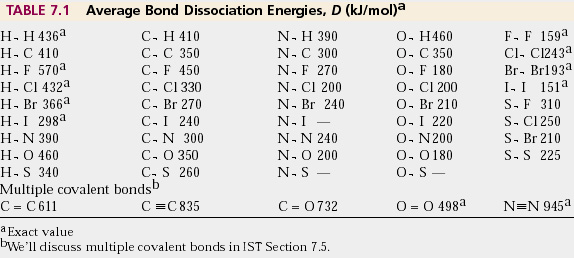
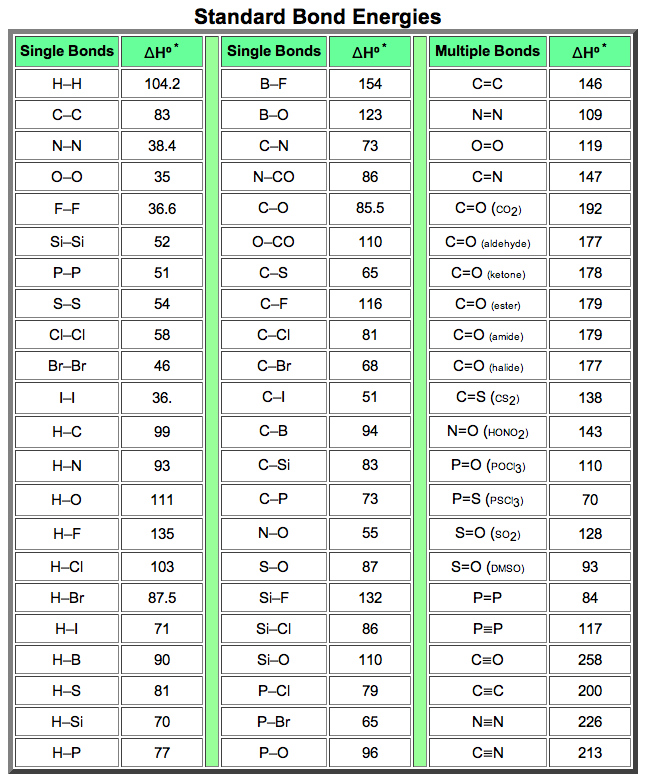
SOLVED: TABLE 9.3 Average Bond Energies Bond Energy (kJ/mol) Bond 436 Bond 414 Bond 389 Bond Energy (kJ/mol) Bond 237 Bond 218 Bond 193 Bond 208 Bond 175 Bond 151 Bond 228
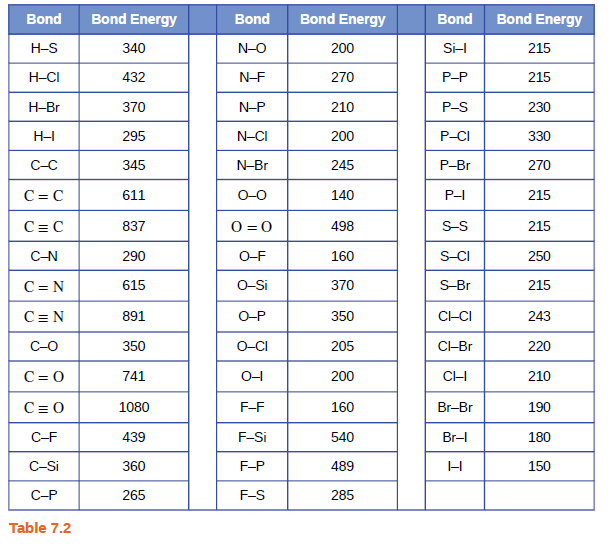
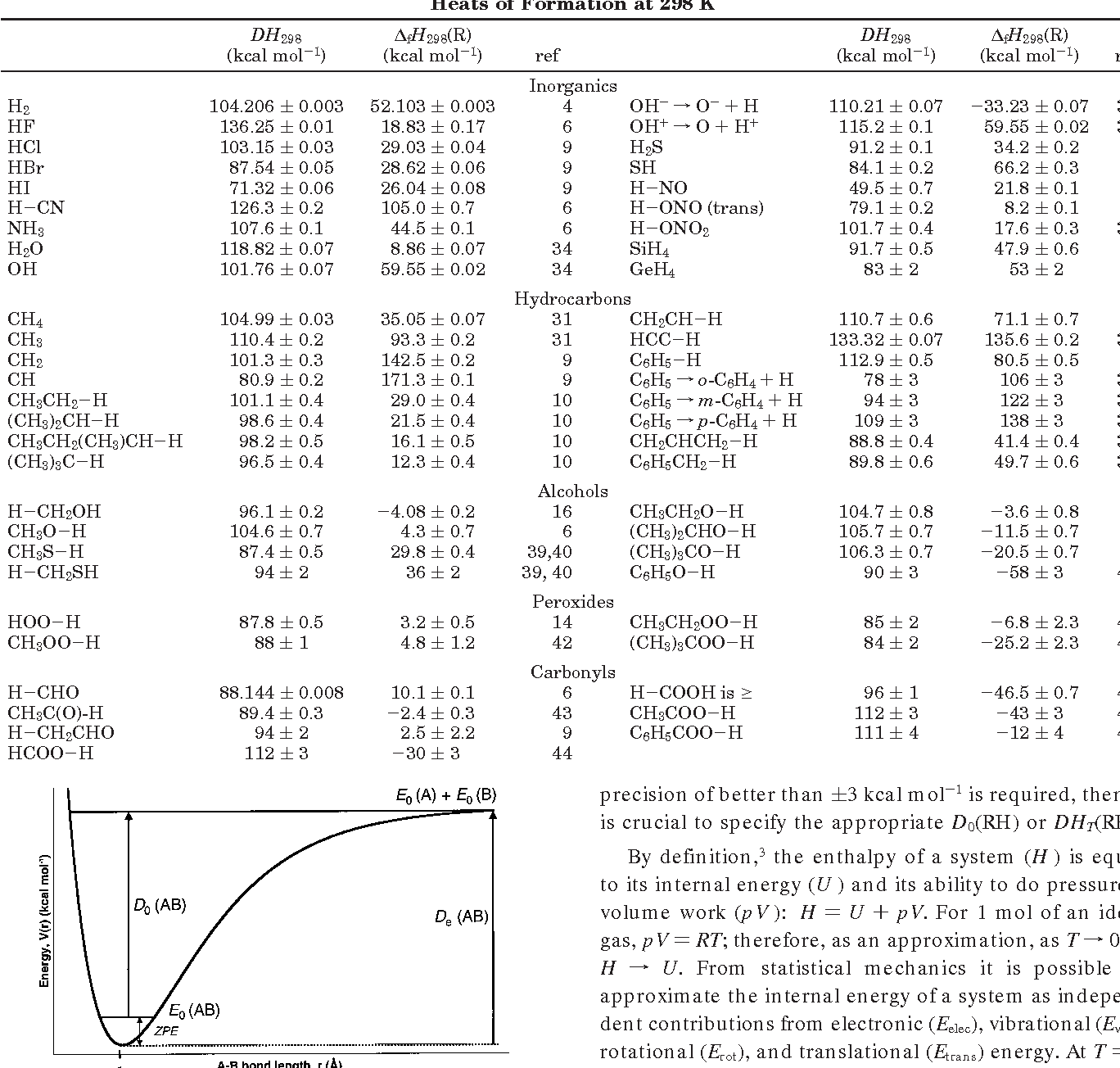
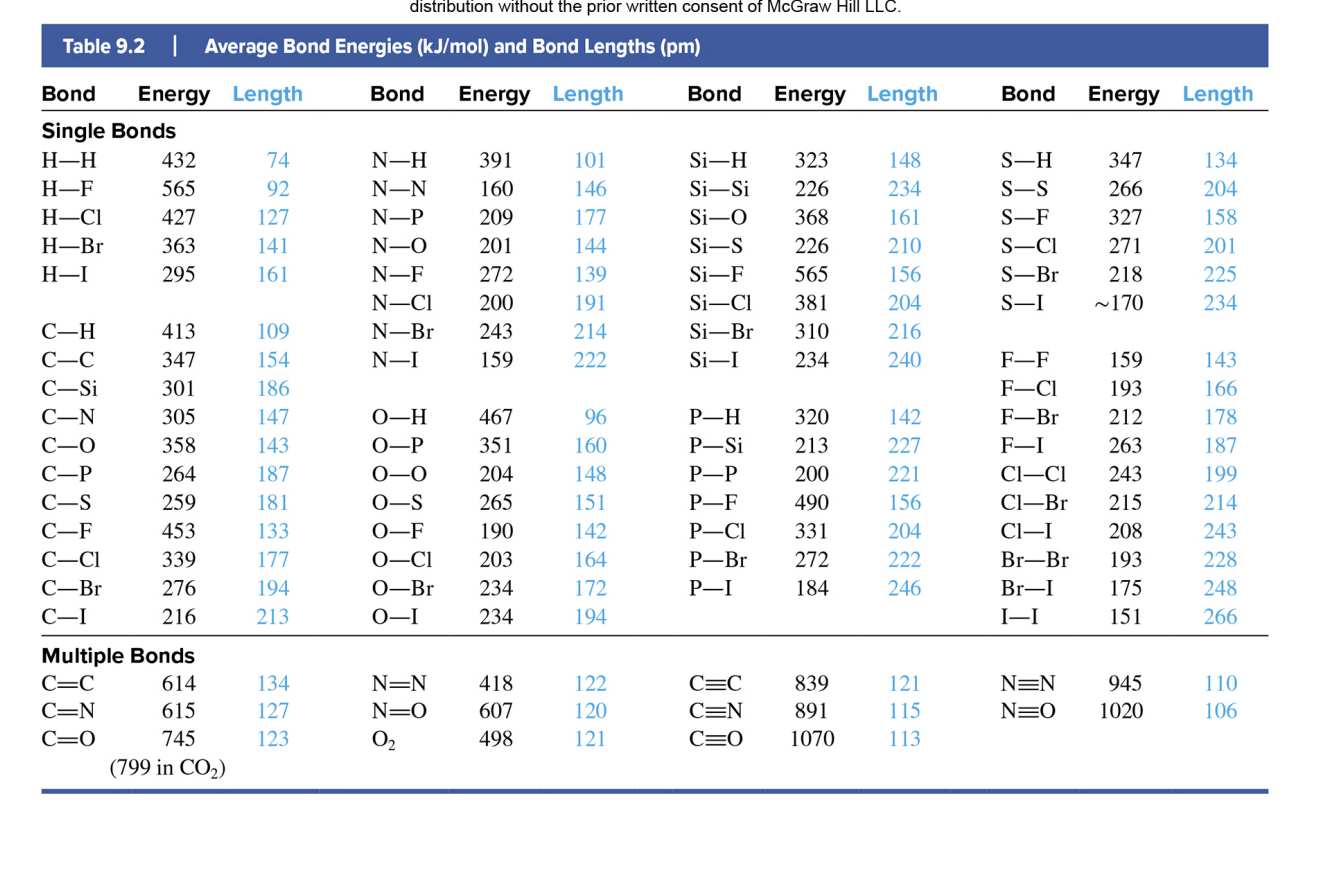
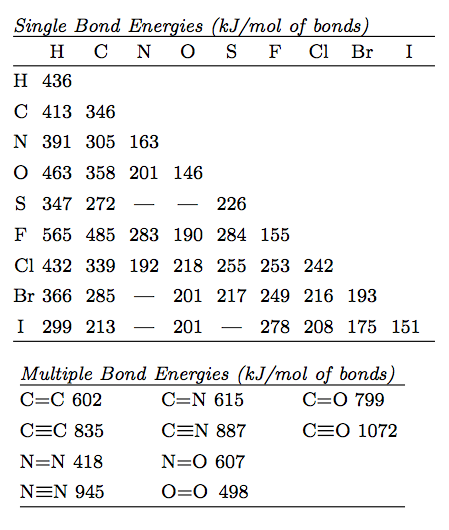
Using the bond energies in Table 7.2, determine the approximate enthalpy change for each of the following reactions: (a) Cl 2 ( g ) + 3 F 2 ( g ) →

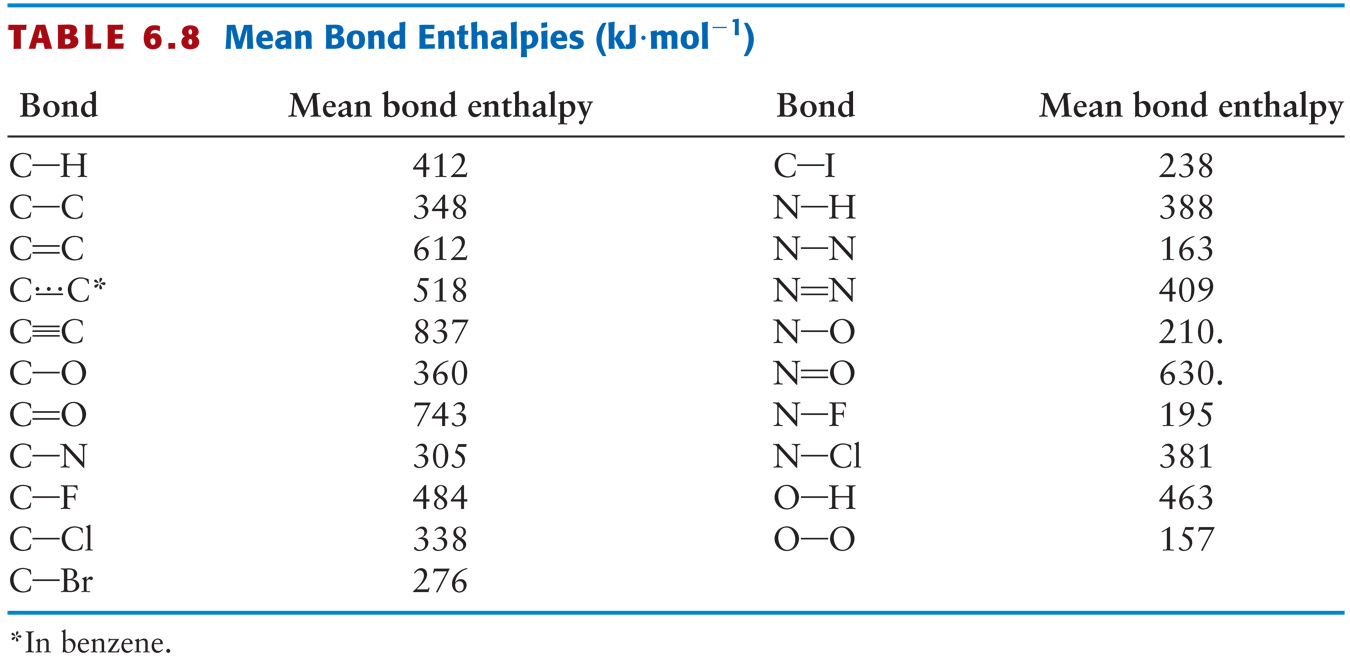
SOLVED: TABLE 9.4 Bond Energies (kJ/mol) Single Bonds C-H 414 C-C 348 C-N 293 C-O 358 C-F 439 C-Cl 328 C-Br 276 C-I 238 C-S 250 N-H 389 N-S 163 N-O 201


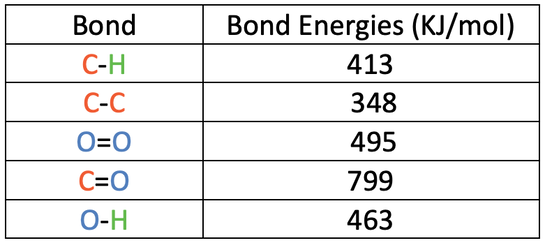








![PDF] Bond dissociation energies | Semantic Scholar PDF] Bond dissociation energies | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ce7c065d005590afbab6bb30de466e75e50d4f86/7-Table3-1.png)