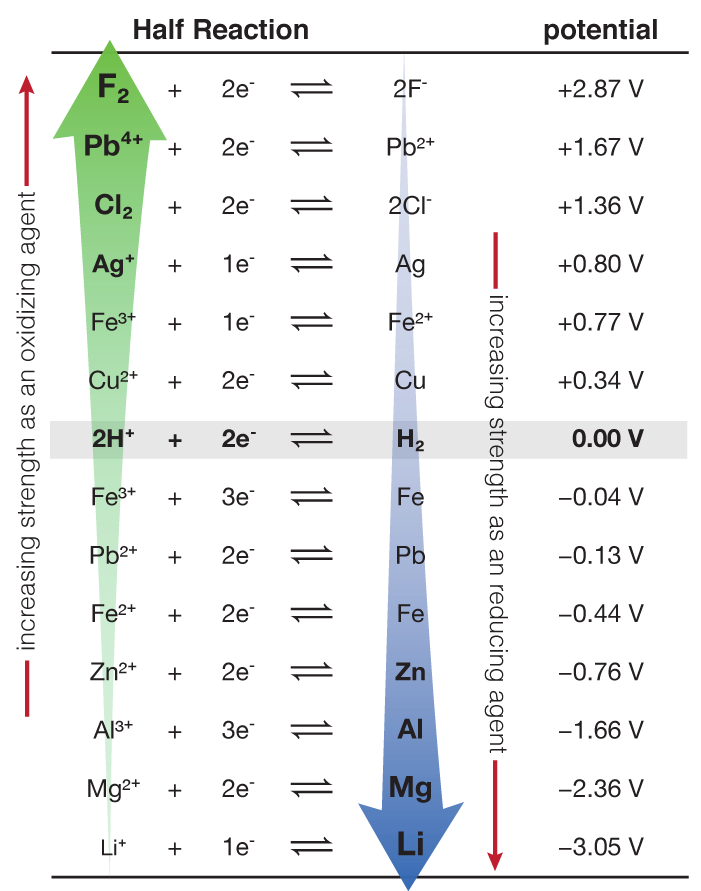
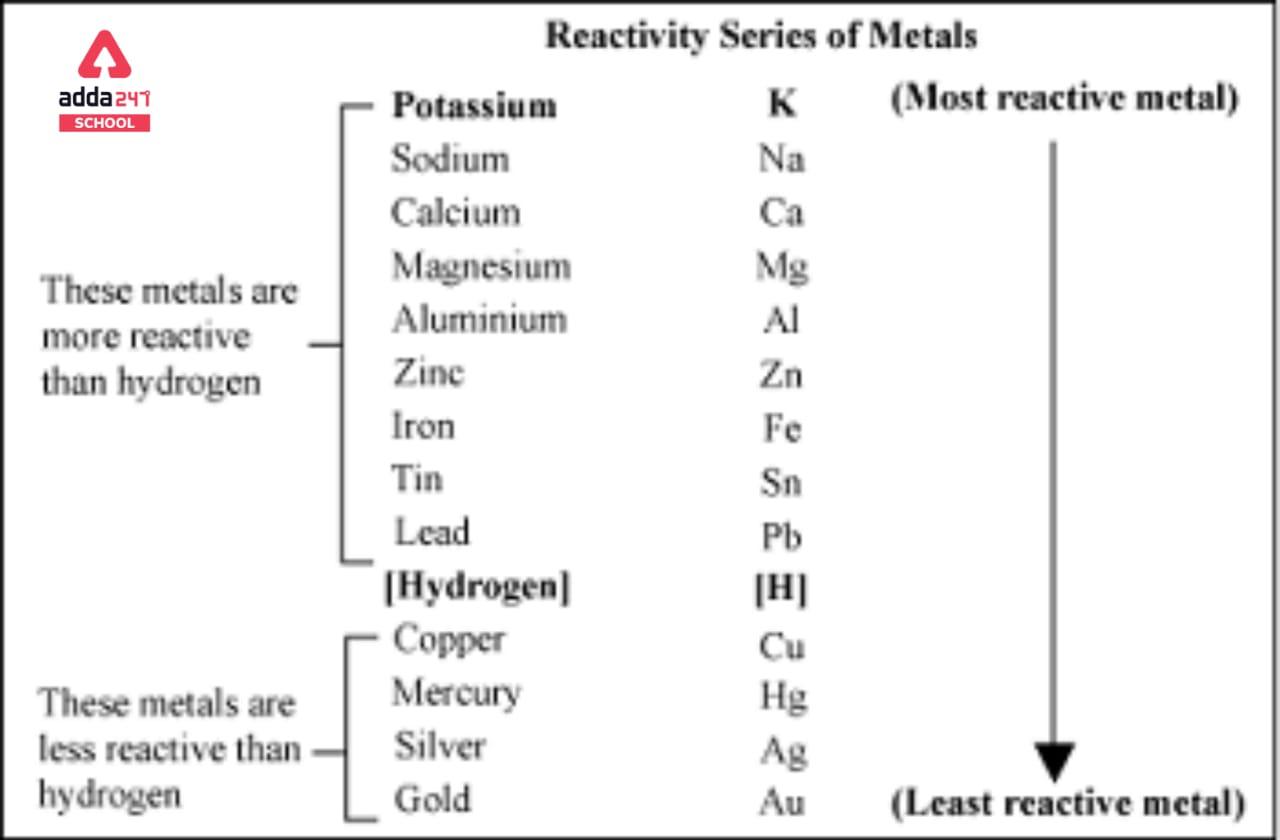
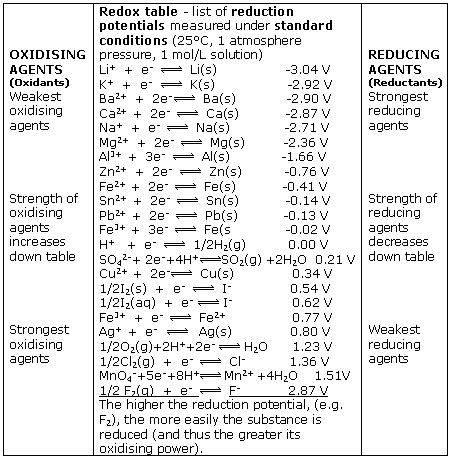
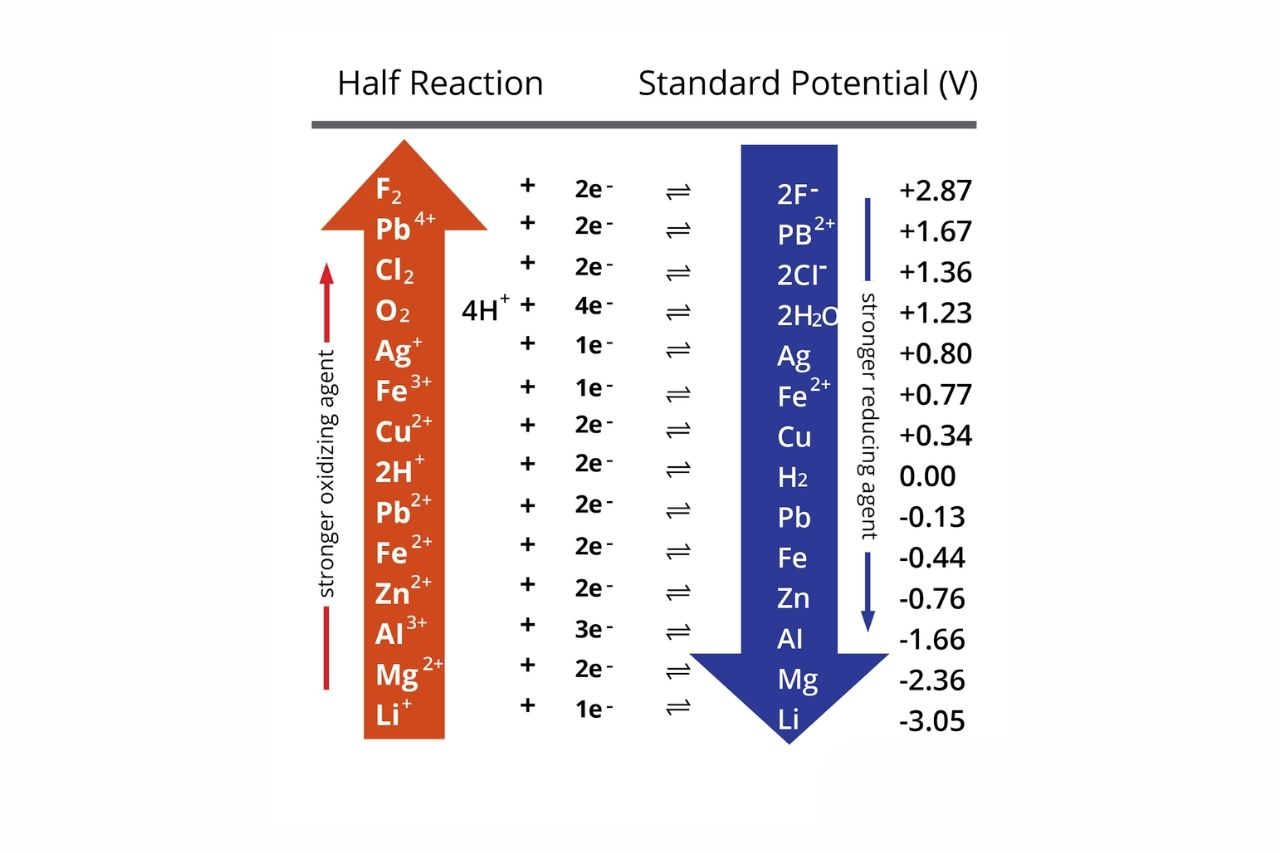
Using the standard electrode potentials given in Table 3.1, predict the reaction between the following is feasible:(i) Fe^{3+}(aq) and I^{-}(aq)(ii) Ag^{+} (aq) and Cu(s)(iii) Fe^{3+} (aq) and Br^{-} (aq)(iv) Ag(s) and Fe^{3+} (

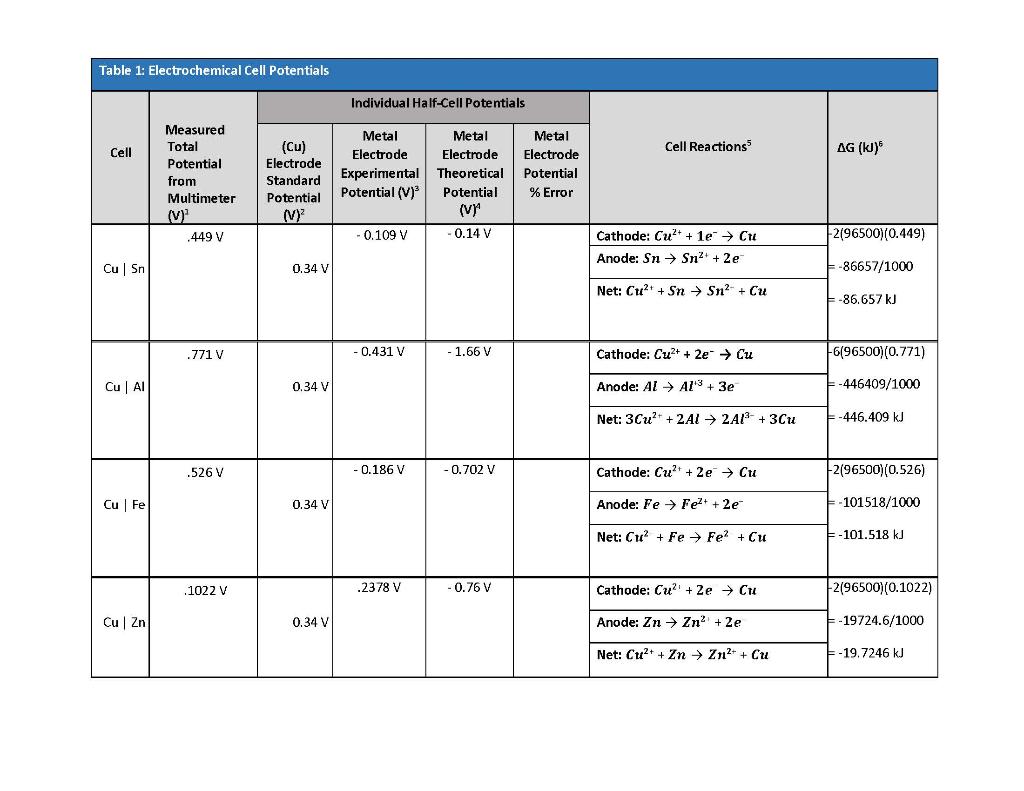
SOLVED: Table 1: Electrochemical Cell Potentials Individual Half-Cell Potentials Measured Total Potential from Multimeter Metal Metal Electrode Electrode Experimental Theoretical Potential (V)? Potential (vn Metal Electrode Potential % Error Cell Cu ...




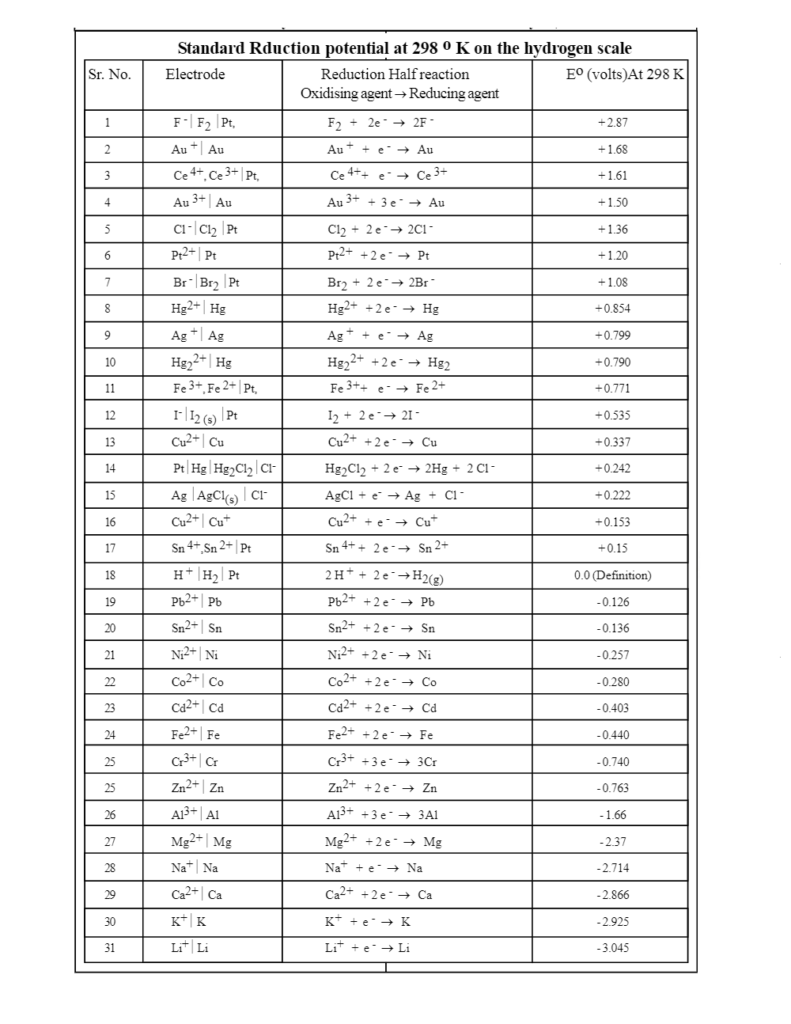
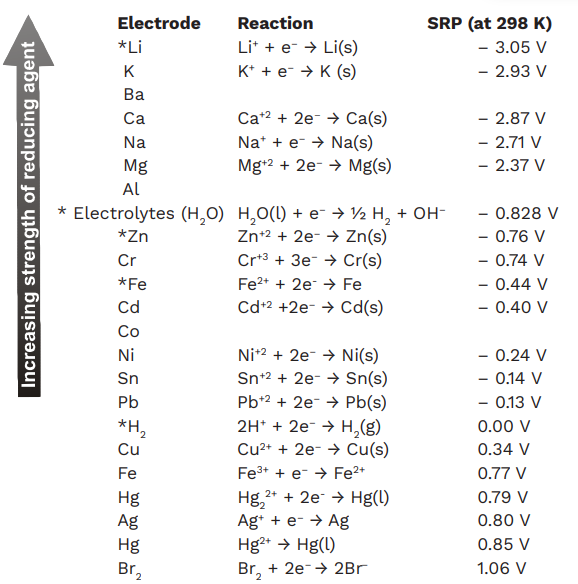








![The variation of the difference of the electrochemical potential [mV]... | Download Table The variation of the difference of the electrochemical potential [mV]... | Download Table](https://www.researchgate.net/publication/279312214/figure/tbl2/AS:667839455326213@1536236742592/The-variation-of-the-difference-of-the-electrochemical-potential-mV-between-the-coat.png)