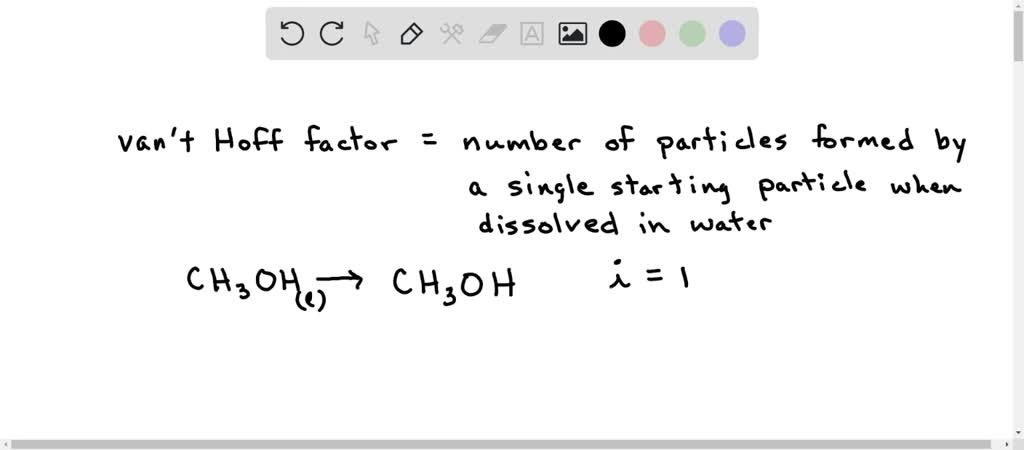128. Which one has same Van't Hoff factor i as that of Hg2Cl2: (1) NaCl (3) Al(NO3)3 (2) Na2SO4(4) Al,(SO4)3
![In which of the following case, van't hoff factor is greater than 3 (A) mathrm{AlCl}_{3} alpha=0.8 (B) mathrm{BaCl}_{2} alpha=0.9 (c) mathrm{NaCl} alpha=0.9 (D) mathrm{K}_{4}left[mathrm{Fe}(mathrm{CN})_{6}right] alpha=0.5 In which of the following case, van't hoff factor is greater than 3 (A) mathrm{AlCl}_{3} alpha=0.8 (B) mathrm{BaCl}_{2} alpha=0.9 (c) mathrm{NaCl} alpha=0.9 (D) mathrm{K}_{4}left[mathrm{Fe}(mathrm{CN})_{6}right] alpha=0.5](https://toppr-doubts-media.s3.amazonaws.com/images/4390395/f7a7e9bd-2451-436d-b371-2afa82a2f7d0.jpg)
In which of the following case, van't hoff factor is greater than 3 (A) mathrm{AlCl}_{3} alpha=0.8 (B) mathrm{BaCl}_{2} alpha=0.9 (c) mathrm{NaCl} alpha=0.9 (D) mathrm{K}_{4}left[mathrm{Fe}(mathrm{CN})_{6}right] alpha=0.5

The Van't Hoff factor of 0.1 M {text{Ba}}{left( {{text{N}}{{text{O}}_3}} right)_2} solutions is 2.74. The degree of dissociation will be 91.3%74%87%100%

For an aqueous solution of HF, determine the van't Hoff factor assuming - Home Work Help - Learn CBSE Forum