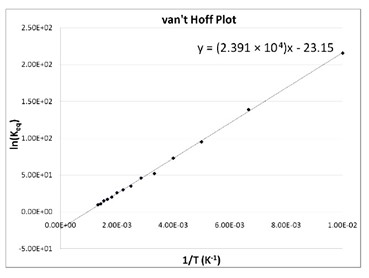
SOLVED: TC = g C o RT van't Hoff equation Osmotic pressure (atm) of particles per mole solution (Osm mol) Concentration (mmol) Reflection coefficient (varies) from Gas constant 0.082 atm mol Absolute

PDF) Van't Hoff equation-equilibrium How much Van't Hoff equation -effect on temperature | Kajal Panda - Academia.edu
ANALOGY BETWEEN ARRHENIUS AND VAN'T HOFF EQUATION :— {Topic: Chemical Kinetics} Arrhenius equation and Van't Hoff equation are two important and often asked about equations in chemistry. Remembering one of these can



![Odia] Define Van't Hoff laws of Osmotic pressure. Odia] Define Van't Hoff laws of Osmotic pressure.](https://static.doubtnut.com/ss/web/11406785.webp)








![D] Laws of Osmotic Pressure and van't Hoff's' Equation van't Hoff (1877).. D] Laws of Osmotic Pressure and van't Hoff's' Equation van't Hoff (1877)..](https://classroom-images.cdn.askfilo.com/classroom/1668316067828_fpynsrpl_3026990.jpg)